Competition of Nucleophilic Aromatic Substitution, σ‐Bond Metathesis, and syn Hydrometalation in Titanium(III)‐Catalyzed Hydrodefluorination of Arenes - Krüger - 2016 - Chemistry – An Asian Journal - Wiley Online Library

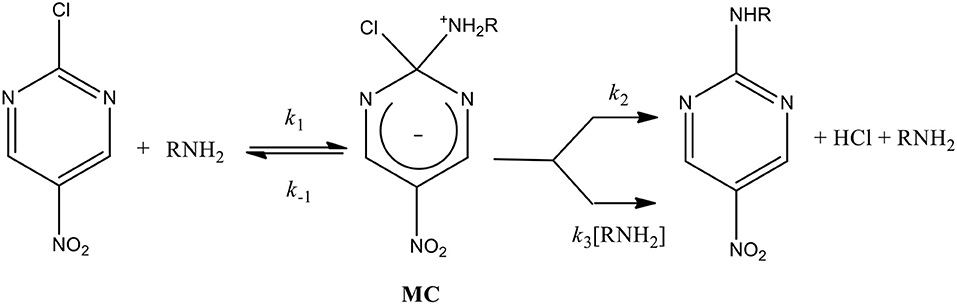
Sulfite-Catalyzed Nucleophilic Substitution Reactions with Thiamin and Analogous Pyrimidine Donors Proceed via an SNAE Mechanism | The Journal of Organic Chemistry

First-principles study of the reaction mechanism governing the SNAr of the dimethylamine on 2-methoxy-5-nitrothiophenes | SpringerLink
Studies on the Synthesis of Perfluoroaryl Sulfides and their Application in Desulfurative Nickel-Catalyzed Reductive Cross- Coup
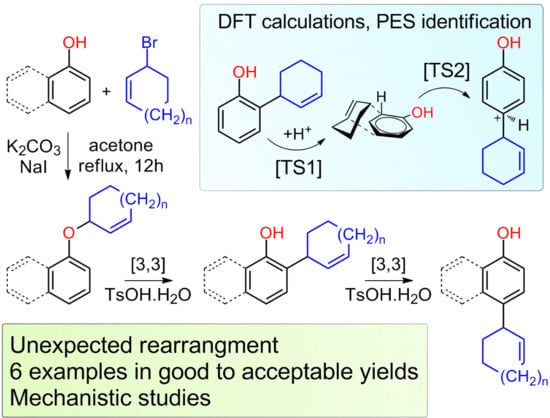
Synthesis of perfluoroaryl sulfides at electron-poor arenes via an SNAr step with an unexpected mechanism
Frontiers | Experimental Analyses Emphasize the Stability of the Meisenheimer Complex in a SNAr Reaction Toward Trends in Reaction Pathways

SNAr Regioselectivity Predictions: Machine Learning Triggering DFT Reaction Modeling through Statistical Threshold | Journal of Chemical Information and Modeling

Synthesis of a novel farnesyl transferase inhibitor, ABT-100; selective preparation of a stereogenic tertiary carbinol - ScienceDirect

SNAr Regioselectivity Predictions: Machine Learning Triggering DFT Reaction Modeling through Statistical Threshold | Journal of Chemical Information and Modeling
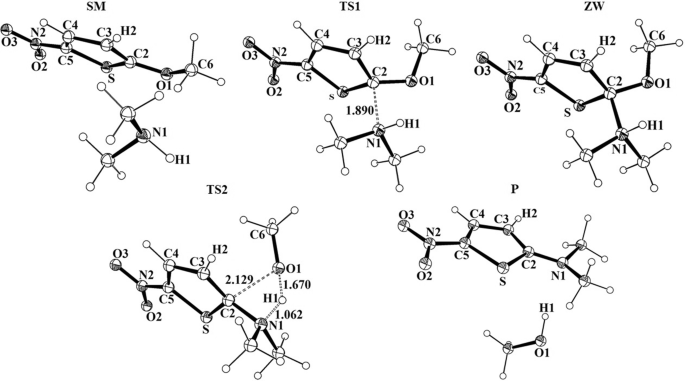
First-principles study of the reaction mechanism governing the SNAr of the dimethylamine on 2-methoxy-5-nitrothiophenes | SpringerLink

Secreted Reporter Assay Enables Quantitative and Longitudinal Monitoring of Neuronal Activity | eNeuro

Automated Transition State Search and Its Application to Diverse Types of Organic Reactions | Journal of Chemical Theory and Computation

Molecules | Free Full-Text | How Do Aromatic Nitro Compounds React with Nucleophiles? Theoretical Description Using Aromaticity, Nucleophilicity and Electrophilicity Indices

Secreted reporter assay enables quantitative and longitudinal monitoring of neuronal activity | bioRxiv

Reaction mechanism of the SNAr reaction between 1-X-2,4-dinitrobenzene... | Download Scientific Diagram
SNAr Regioselectivity Predictions: Machine Learning Triggering DFT Re- action Modeling through Statistical Threshold

SNAr Regioselectivity Predictions: Machine Learning Triggering DFT Reaction Modeling through Statistical Threshold | Journal of Chemical Information and Modeling

Synthesis of perfluoroaryl sulfides at electron-poor arenes via an SNAr step with an unexpected mechanism - ScienceDirect

Reaction mechanism of the SNAr reaction between 1-X-2,4-dinitrobenzene... | Download Scientific Diagram